Ultrasonic process
Cleaning in most instances requires that a contaminant be dissolved (as in the case of a soluble soil), displaced (as in the case of a non-soluble soil) or both dissolved and displaced (as in the case of insoluble particles being held by a soluble binder such as oil or grease). The mechanical effect of ultrasonic energy can be helpful in both speeding dissolution and displacing particles. Just as it is beneficial in cleaning, ultrasonics is also beneficial in the rinsing process. Residual cleaning chemicals are removed quickly and completely by ultrasonic rinsing.
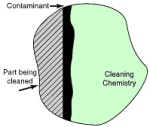
In removing a contaminant by dissolution, it is necessary for the cleaning chemistry to come into contact with and dissolve the contaminant. The cleaning activity takes place only at the interface between the cleaning chemistry and the contaminant. As the cleaning chemistry dissolves the contaminant, a saturated layer develops at the interface between the fresh cleaning chemistry and the contaminant. Once this has happened, cleaning action stops as the saturated chemistry can no longer attack the contaminant.
Fresh chemistry cannot reach the contaminant
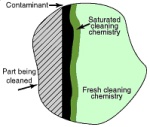
Ultrasonic cavitation and implosion effectively displace the saturated layer to allow fresh chemistry to come into contact with the contaminant remaining to be removed. This is especially beneficial when irregular surfaces or internal passageways are to be cleaned.
 |
 |
 |
Please raise your question
VARICLEAN benelux bv is specialist in ultrasonic cleaning for industrial and professional applications. On base of your cleaning requirement we would be pleased to provide you the right advise and an adequate solution.

